Theories of Osmosis
Several theories have been developed to explain the action of a semipermeable membrane. It is probable that the mechanism depends on the particular type of membrane used and also on the nature of the solute and solvent. Some of the more important theories of osmosis across a membrane are discussed below-
The Molecular Sieve Theory
According to this theory, the membrane contains lots of fine pores and acts as a sort of molecular sieve. Smaller solvent molecules can pass through the pores but the larger molecules cannot. Solvent molecules flow from a region of higher solute concentration to one of lower concentration across such a membrane. But since some membranes can act as sieves even though the solute molecules are smaller than the solvent molecules, this theory remains in doubt.
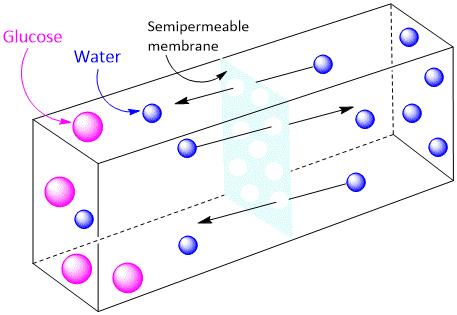
Recently it has been shown that the pores or capillaries between the protein molecules constituting an animal membrane are lined with polar groups (–COO–, –NH3+, –S–2, etc.). Therefore, the membrane acts not simply as a sieve but also regulates the passage of solute molecules by electrostatic or chemical interactions. In this way even solute molecules smaller than solvent molecules can be held back by the membrane.
Membrane Solution Theory
Membrane proteins bearing functional groups such as – COOH, –OH, –NH2, etc., dissolve water molecules by hydrogen bonding or chemical interaction. Thus membrane dissolves water from the pure water (solvent) forming what may be called ‘membrane solution’. The dissolved water flows into the solution across the membrane in a bid to equalise concentrations. In this way water molecules pass through the membrane while solute molecules being insoluble in the membrane do not.
Vapour Pressure Theory
It suggests that a semipermeable membrane has many fine holes or capillaries. The walls of these capillaries are not wetted by water (solvent) or solution. Thus neither solution nor water can enter the capillaries. Therefore each capillary will have in it solution at one end and water at the other, separated by a small gap.
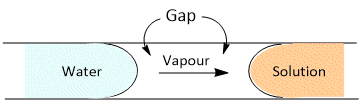
Since the vapour pressure of a solution is lower than that of the pure solvent, the diffusion of vapour will occur across the gap from water side to solution side. This will result in the transfer of water into the solution. The vapour theory offers a satisfactory explanation of the mechanism of osmosis in most cases.
Membrane Bombardment Theory
This theory suggests that osmosis results from an unequal bombardment pressure caused by solvent molecules on the two sides of the semipermeable membrane. On one side we have only solvent molecules while on the other side there are solute molecules occupying some of the surface area. Thus there are fewer bombardments per unit area of surface on the solution side than on the solvent side. Hence the solvent molecules will diffuse more slowly through the membrane on the solution side than on the solvent side. The net result causes a flow of the solvent from the pure solvent to the solution across the membrane.
